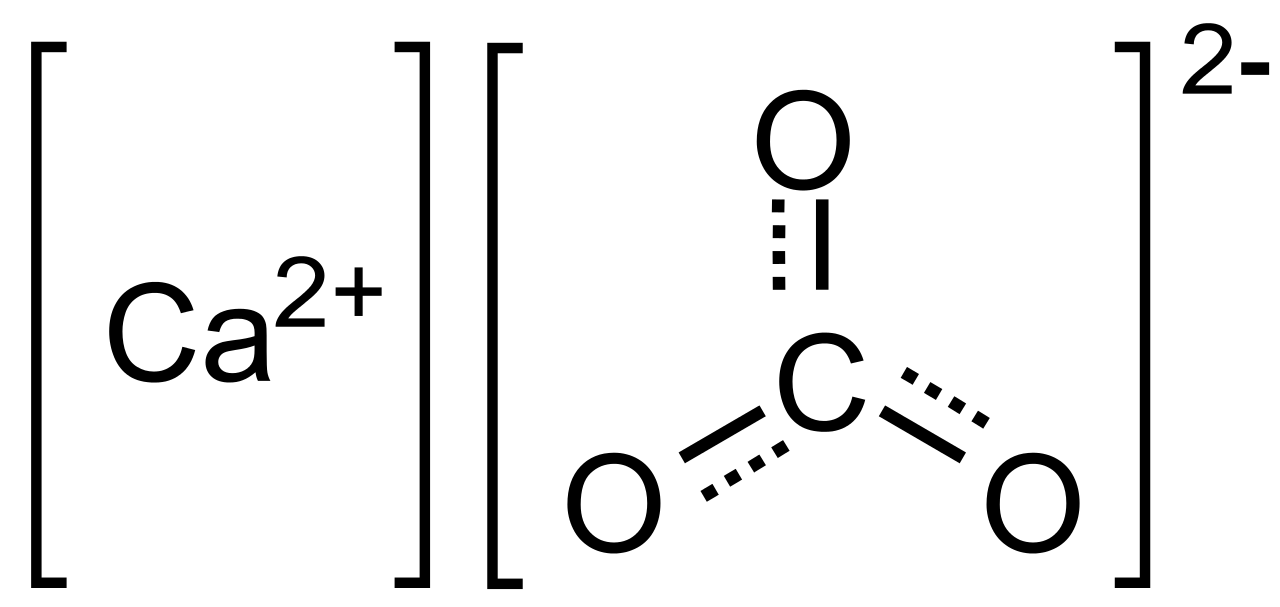
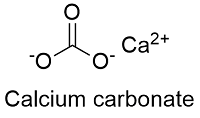
Calcium carbonate equilibrium or saturation (SI = 0, at 25 ◦ C and {HCO... | Download Scientific Diagram

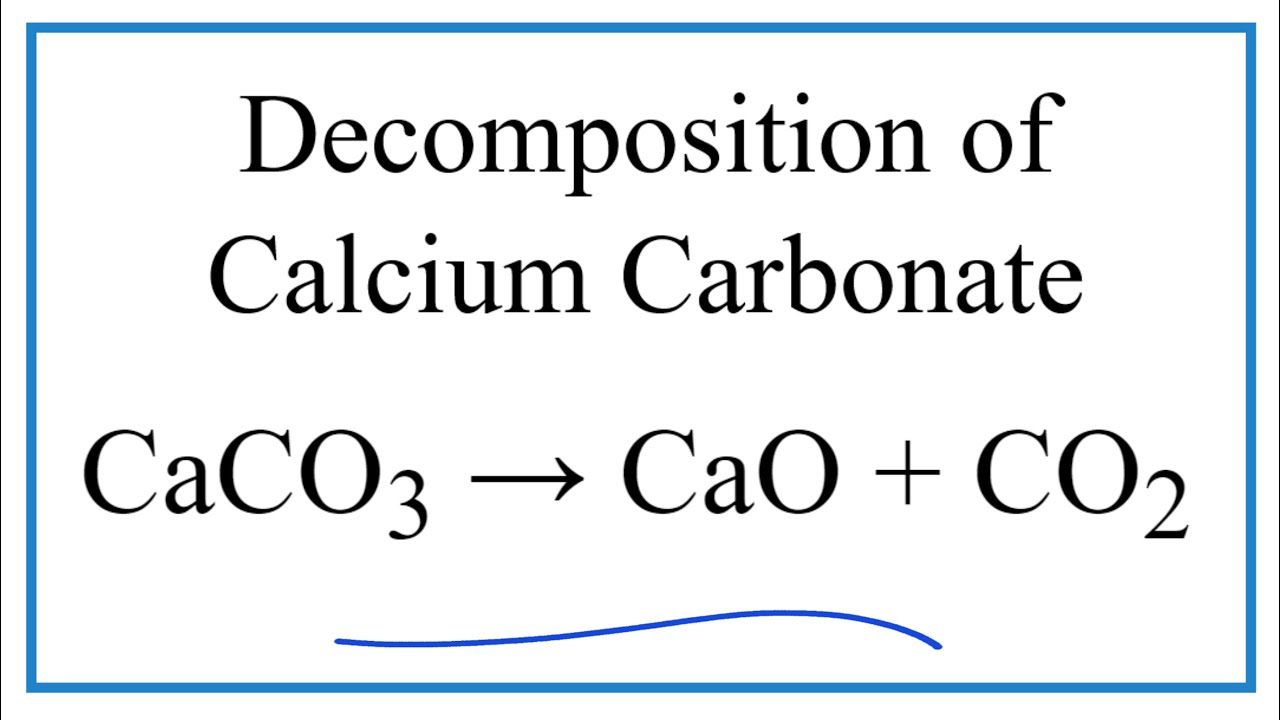
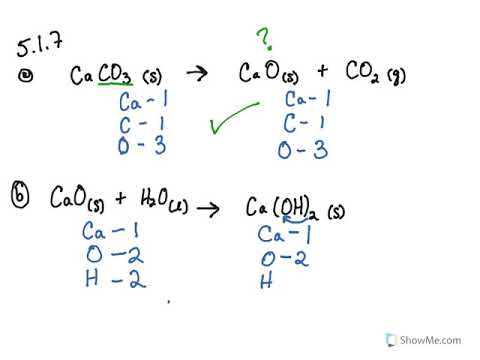
Balance the equation in which, calcium carbonate decomposes, on heating, to form calcium oxide and carbon dioxide
Write balanced chemical equation for the following processes: (a) heating calcium in oxygen (b) heating calcium carbonate - Sarthaks eConnect | Largest Online Education Community

Write a balanced chemical equation for the reaction of calcium carbonate and dil. hydrochloric acid.
What is the Ionic equation for the reaction between calcium carbonate and hydrochloric acid? - Quora

Question Video: Calculating the Mass of Calcium Carbonate Required to Produce a Given Mass of Calcium Oxide | Nagwa

Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride