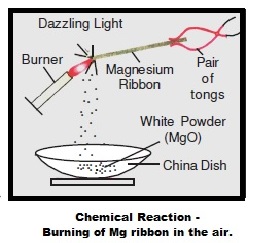
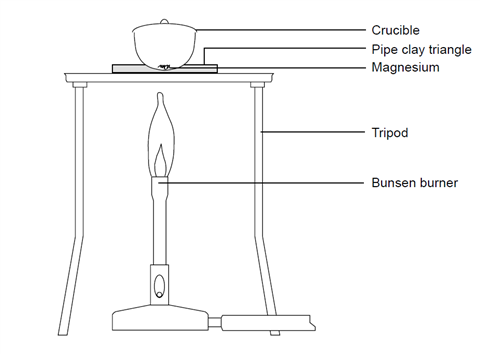
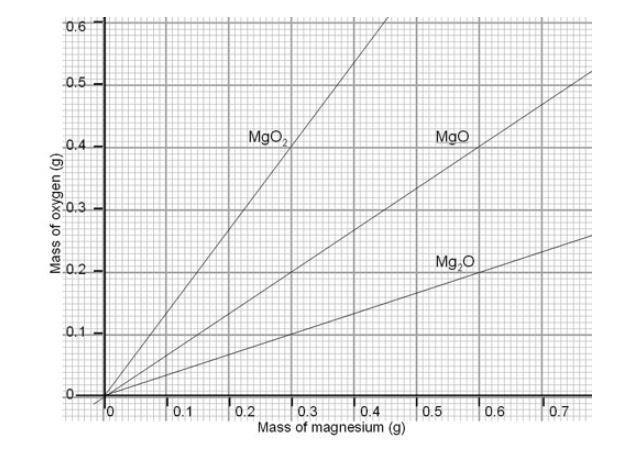
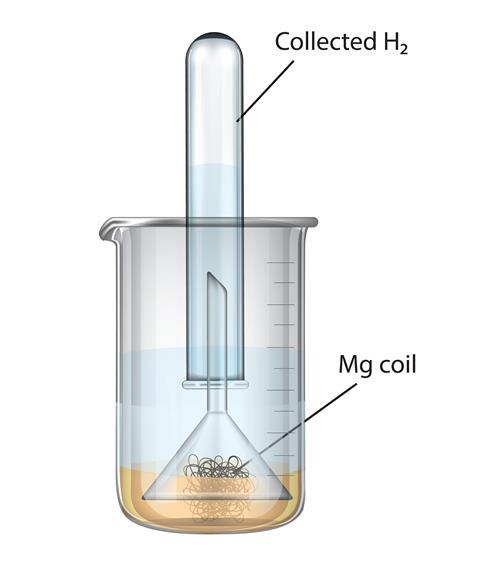
A magnesium ribbon is burnt in oxygen to give a white compound X accompanied by emission of light. If the burning ribbon is now placed in an atmosphere of nitrogen, it continues

Chemical equation: A set of symbols and chemical formula is representing the reactants and products molecules in the chemical reaction and the conditions. - ppt download

SOLVED: Burning magnesium ribbon Magnesium ribbon (which; like most metals looks silver and shiny) bumns in the presence of oxygen gas in a combination reaction Write the coirect product(s) of this reaction

Magnesium ribbon burns with a dazzling flame in air (or oxygen) and changes to white substance magnesium oxide. Is magnesium being oxidised or reduced in this reaction ?

Chemical Equations. When chemicals are added together and a reaction occurs a chemical equation can be written The reactants go on the left of the equation. - ppt download

Whan a burning magnesium ribbon is introduced in a gas jar containing oxygen, it will burn with : - YouTube

A student performs an experiment of burning magnesium ribbon in the air. A chemical reaction takes place and as a result, a white powder X forms along with a bright white light.The