Write the balanced chemical equations for the folowing reactions (a) Calcium hydroxide + Carbon - YouTube

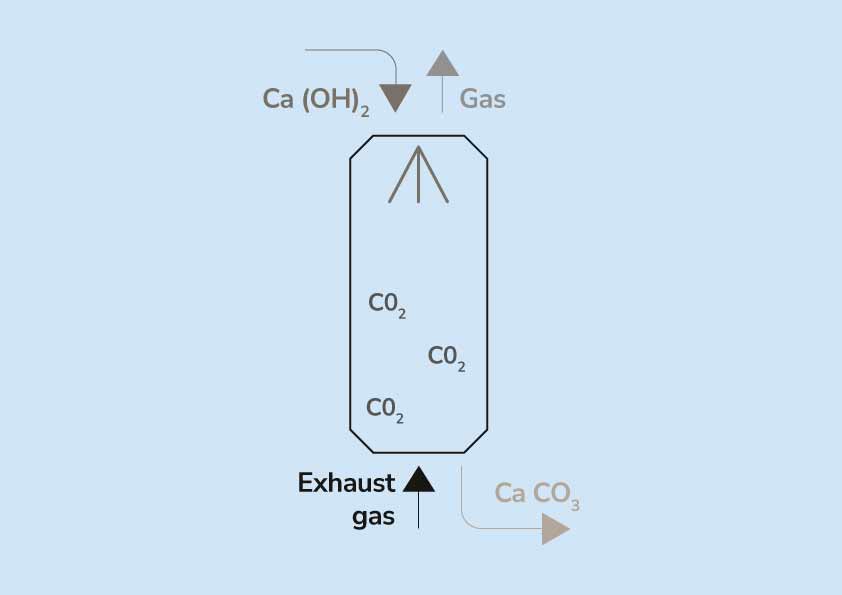
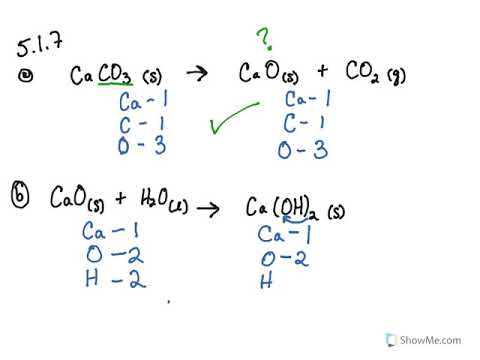
Write the balanced chemical equations for the following reaction:Calcium hydroxide + Carbon dioxide → Calcium Carbonate + Water.

AQA GCSE Science & Additional Science Chemistry 1 Topic 2 Hodder Education Revision Lessons Limestone and building materials Click to continue. - ppt download

Look at Figure 4.1 and answer the following questions (a) What change would you observe in the calcium hydroxide solution taken - Question 53, 4. Carbon and its Compounds, Ncert Exemplar -

Reactions of carbon dioxide - Gas chemistry - (CCEA) - GCSE Chemistry (Single Science) Revision - CCEA - BBC Bitesize

science chemistry precipitation reaction carbon dioxide | Fundamental Photographs - The Art of Science
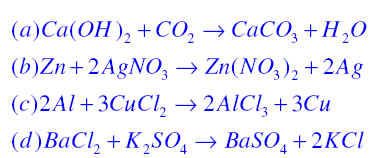
Write the balanced chemical equations for the following reactions. (a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water | Pushpender86's Blog

Ca(OH)2+CO2 =CaCO3 +H2O Balanced Equation|Calcium hydroxide+Carbon dioxide=Calcium carbonate + Water - YouTube